Our Approach
Everything we do at PFIC Network starts with the people living with PFIC: patients, parents, family members, and caregivers who navigate this disease every day. Their experiences, priorities, and unmet needs are the foundation of our advocacy and research efforts.
From 2020 to 2023, PFIC Network was a grantee of the Chan Zuckerberg Initiative’s Rare As One program, which supported us in building a patient-led collaborative research network for PFIC. That investment helped establish the infrastructure behind many of our initiatives — including our patient registry, the PFIC Family & Scientific Conference, and a framework for bringing together patients, clinicians, and researchers across borders to collaborate on shared priorities.
Key Priorities
Through the projects highlighted below, we are working to systematically capture and share what life with PFIC is really like and bring those voices directly to regulatory agencies, researchers, and those working to develop treatments for PFIC.
The insights we gather help us focus our efforts on three key priorities:
Stronger Together
What makes these efforts especially powerful is how they build on one another. Registry data reveals the scope of unmet needs, research initiatives like IMPACT translate those needs into defined priorities, and direct engagement with people living with PFIC and their families adds depth and context that numbers alone can’t capture.
Together, they give us a stronger, more credible voice when advocating for change.
Explore our initiatives
Below, you can explore the projects and initiatives that drive our community-driven advocacy and research efforts. Each section provides a brief overview with links to learn more.
This project, funded by a Eugene Washington Engagement Award (EASO-30455) from the Patient-Centered Outcomes Research Institute (PCORI), was designed to build capacity for patient-centered comparative clinical effectiveness research (CER) in PFIC. For two years, PFIC patients and caregivers worked alongside clinicians and researchers to:
- Design tools to support planning for future patient-centered CER
- Identify CER targets
- Define what it means to collaborate in research projects.
These efforts produced two ongoing resources: the IMPACT Research Roadmap and the Treatment Experience App.

A planning tool and resource library reflecting the shared insights and contributions of PFIC patients, parents, clinicians, and researchers. Its purpose is to foster sustainable multi-stakeholder collaboration and guide the development of future patient-centered CER projects in PFIC.
It also provides additional details about Project IMPACT, including the project’s motivation, structure, and methodology — as well as companion materials for researchers looking to build and sustain engagement with the PFIC community or design a PFIC CER study around patient-defined priorities.
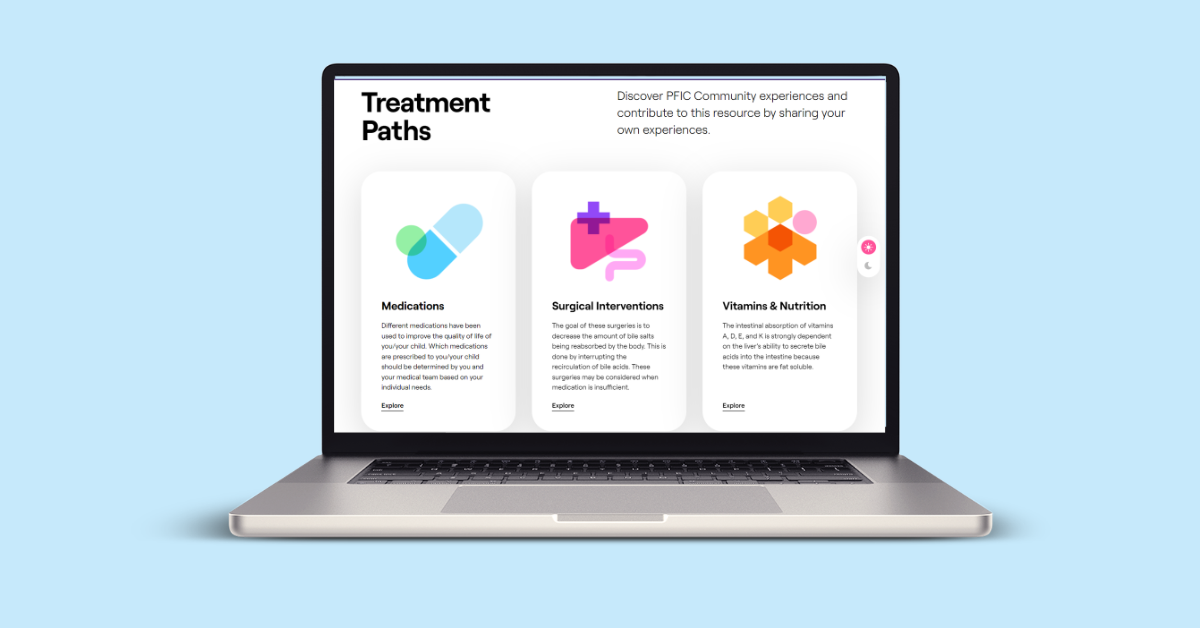
The TEA is an interactive web platform designed by PFIC patients, families clinicians, and researchers in IMPACT to:
- Provide information about PFIC treatments and patients’ experiences with them
- Learn which treatment outcomes matter most so that we can start to design research studies that are directly relevant to the PFIC community
It is a living resource, updated with summaries of patient responses to the in-app survey.

2022- Ongoing, CZI Rare As One
PFIC Network Patient Registry (PNPR)
The PFIC Network Patient Registry (PNPR) is a secure, longitudinal data collection where patients or their caregivers report information about their experience with the disease.
- Collects data on demographics, diagnosis, symptoms, medications, and surgeries
- Uses psychometrically validated questionnaires to assess itch, sleep, global health, family quality of life, and financial burden
- Participants complete follow-up surveys every six months to track changes over time
Baseline data from the PNPR has been published in a peer-reviewed paper in collaboration with research partners, and preliminary findings have been presented in several posters at medical conferences. Researchers may request de-identified registry data or contact participants who have opted in to learn about research opportunities.

PFIC families around the world face vastly different challenges depending on where they live. Project REACH (Research, Education, Advocacy, Collaboration, and Hope) was our initiative to understand the unique barriers facing PFIC patients in underserved communities and turn those insights into action.
Funded by a Global Genes Health Equity in RARE Patient Impact grant, REACH used workshops and surveys to bring together patients and healthcare providers, assess and prioritize unmet needs, and develop country-specific advocacy action plans for PFIC communities in India and Pakistan.

In March 2021, PFIC Network participated in a virtual FDA Patient Listening Session, a small, nonpublic discussion where patients and caregivers share their experiences living with a disease directly with FDA staff. During the session, PFIC parents shared firsthand accounts of the daily burden of pruritus, the limitations of current treatment options, and the complications families face before and after liver transplant.
A clinical perspective on disease burden and unmet treatment needs was also presented. The FDA uses insights from listening sessions to inform guidance documents, medical product development, and regulatory decision-making.
What’s Next?
Much of the work highlighted above is ongoing. Sign up for our mailing list to find out about the latest project updates and new initiatives!